North Carolina Tissue Consortium
Mission

The North Carolina Tissue Consortium (NCTC), an IRB-approved tissue bank, is supported by the University Cancer Research Fund which was established by the NC General Assembly to accelerate the battle against cancer. The objective of the NCTC is to facilitate cancer-related research by providing a means through which normal and malignant tissue specimens are procured, processed, stored, and distributed to researchers while protecting the rights and confidentiality of participants. We also provide consultations on an as-needed basis to help identify and target specimen collection to meet the specific research needs of investigators.
Contact Information
Please contact the NCTC, NCTC Director Kathryn Verbanac, PhD or Research Specialist Iyana Beachem to discuss any questions you may have related to specific research needs.

Kathryn M. Verbanac
Professor, Division of Surgical Oncology
Department of Surgery
4S-16B Brody School of Medicine
verbanack@ecu.edu
(252)744-3689

Iyana M. Beachem
Research Specialist, Division of Surgical Oncology
Department of Surgery
4W-23 Brody School of Medicine
beachemi25@ecu.edu
(252)744-5879
Informed Consent and Privacy
Patients with or suspected of having cancer and undergoing a cancer-related surgery or procedure will be presented with the opportunity to participate in this tissue banking study. An informed consent document, approved by the University and Medical Center Institutional Review Board (UMCIRB), will be signed by all patients who elect to donate their tissue to the NCTC.
Collection, Procurement, and Storage
Tissue will only be procured after the amount of specimen needed for diagnostic purposes has been determined by appropriate personnel in the ECU Pathology Department. In addition, H&E-stained slides are evaluated by a board-certified reviewing QC pathologist, to ensure that appropriate and representative diagnostic tissue is procured. Preparations of fresh, snap-frozen, and formalin-fixed paraffin-embedded tissue, and H&E-stained slides are available to researchers. Special processing requests may be fulfilled after consultation with the tissue bank research specialist. Blood or other body fluid samples may also be procured upon request by investigators. Samples may be annotated with patient demographics, such as age or age range, gender, race, height, weight etc. A de-identified pathology report may be provided, if available and requested.
Specimen Request Process
The NCTC is a resource made available primarily to East Carolina University researchers to support cancer related research. De-identified specimens may also be provided to extramural collaborators of ECU faculty (with IRB approval or IRB verification as exempt). Members of the Lineberger Cancer Center at the University of North Carolina at Chapel Hill may also request samples.

- PI contacts NCTC with a project description re: specimen availability.
Contact the NCTC. - PI contacts UMCIRB for project evaluation and IRB determination.
Studies which are determined as exempt or designated as not human subject research do NOT need to be submitted for review on ePirate. Other studies must be submitted for expedited or full UMCIRB review. Contact the UMCIRB. - PI submits Research Agreement/Specimen Request form to NCTC
After PIs submit a NCTC Research Agreement/Specimen Request Form with documentation of the IRB determination, tissues may be dispersed. A separate Request and Research Agreement form and IRB determination will need to be submitted for each individual study and if additional specimens and/or information are requested that were not included in the original request. Contact the NCTC.
Specimens Available (malignant tissue and adjacent uninvolved tissue)
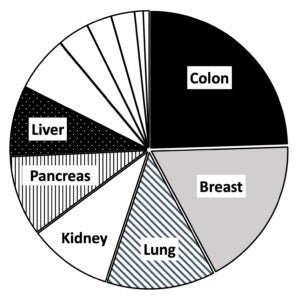
The pie chart below shows the major tissues available. Additional available specimens include stomach, ovary, uterus, prostate, brain, and spleen. Please contact the NCTC for current pricing of services offered.
NCTC Tissues
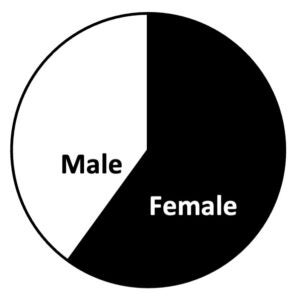
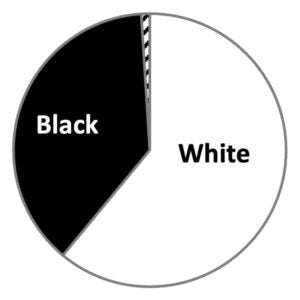
Enrolled Participants
NCTC Support
The NCTC partners and consults with colleagues from Surgery and Pathology.

Nasreen A. Vohra, MD
Associate Professor and Chief,
Division of Surgical Oncology
Department of Surgery
Brody School of Medicine
vohran@ecu.edu

Yaolin Zhou, MD
Associate Professor and Head, Molecular Pathology
Director, Quality/Test Utilization
Department of Pathology and Laboratory Medicine
Brody School of Medicine
zhouy20@ecu.edu
Impact
NCTC clinical specimens have been used by ECU researchers for a variety of studies, leading to significant published insights into cancer signaling pathways, biomarkers, bioenergetics, and potential therapeutic targets. Publications include:
- Fender AW, Nutter JM, Fitzgerald TL, Bertrand FE, Sigounas G. Notch-1 promotes stemness and epithelial to mesenchymal transition in colorectal cancer. J Cell Biochem. 2015;116(11):2517–2527
- Ladin DA, Soliman E, Escobedo R, et al. Synthesis and evaluation of the novel prostamide, 15-deoxy, Δ 12,14 -prostamide J 2 , as a selective antitumor therapeutic. Molecular Cancer Therapeutics. 2017;16(5):838.
- Akula SM, Ruvolo PP, McCubrey JA. TP53/miR-34a-associated signaling targets SERPINE1 expression in human pancreatic cancer. Aging (Albany NY). 2020;12(3):2777–2797.
- Balestrieri K, Kew K, McDaniel M, et al. Proteomic identification of tumor- and metastasis-associated galectin-1 in claudin-low breast cancer. Biochim Biophys Acta Gen Subj. 2021;1865(2):129784.
- McLaughlin KL, Nelson MAM, Coalson HS, et al. Bioenergetic phenotyping of DEN-induced hepatocellular carcinoma reveals a link between adenylate kinase isoform expression and reduced complex I-supported respiration. Front Oncol. 2022;12:919880.
